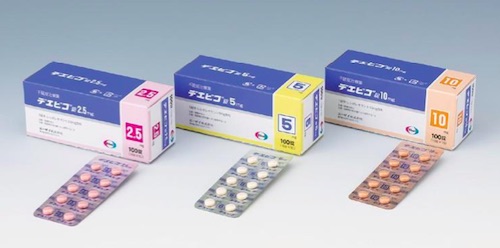
Eisai to Launch In-House Developed New Anti-insomnia Drug Dayvigo (Lemborexant) with Indication for Insomnia in Japan
- Written by ACN Newswire - Press Releases
- Published in ACN Newswire
- Hits: 2226
- Print , Email

|
|
DAYVIGO is a dual orexin receptor antagonist that inhibits orexin neurotransmission regulating sleep-wake rhythm by binding competitively to the two subtypes of orexin receptors (OX1R and OX2R). DAYVIGO acts on the orexin neurotransmitter system and is believed to facilitate sleep onset, sleep maintenance, and wake by regulating sleep-wake rhythm.
The approval of DAYVIGO in Japan is based on findings from two pivotal Phase III studies(1),(2) (SUNRISE 1 and SUNRISE 2) in adult patients with insomnia, as well as evaluation of residual effects including the middle of the night waking, next morning postural stability (falling prediction indicator), and memory through Studies 1063 and 1084.
The SUNRISE 11 clinical trial conducted in North America and Europe utilized objective assessment with overnight measurement through polysomnography, and confirmed statistically significant shortening or improvement of sleep onset latency (primary objective) as well as sleep efficiency and wake after sleep onset (secondary objectives) with DAYVIGO compared to tartrate-sustained-release drug zolpidem (6.25 mg, not yet approved in Japan) and placebo.
The SUNRISE 22 clinical trial, conducted globally including in Japan, evaluated patients subjectively through sleep diaries and confirmed statistically significant improvement in sleep onset latency (primary objective) as well as subjective sleep efficiency and subjective wake after sleep onset (secondary objectives) compared to placebo. Main side effects of DAYVIGO as observed in the two trials were somnolence, headache, dizziness, and fatigue.
Analyses in both studies suggested DAYVIGO was not associated with rebound insomnia, and there was no evidence of withdrawal effects following treatment discontinuation, suggesting it does not produce physical dependence in those taking it for up to one year.In a special safety study (Study 106)(3), DAYVIGO at 5 mg and 10 mg doses did not cause statistically significant impairment in the next morning driving performance in healthy adult or elderly subjects (compared with placebo). Additional special safety studies (Study 108)(4) evaluated middle-of-the-night safety, next morning postural stability, and memory. The effects of DAYVIGO on next day postural stability and memory were evaluated in two randomized, placebo and active-controlled trials. There were no meaningful differences between DAYVIGO and placebo on next-day postural stability or memory at either dose. While there is a need for caution regarding the potential for middle-of-the-night postural instability as well as attention and memory impairment, no problem-signifying degradation was observed between DAYVIGO- and placebo-administered groups.
In June 2020, DAYVIGO was launched in the U.S. for the treatment of adult patients with insomnia, characterized by difficulties with sleep onset and/or sleep maintenance. In addition, Eisai submitted a new drug application seeking approval of DAYVIGO in Canada and Australia.
Insomnia is characterized by difficulty falling asleep, staying asleep, or both, despite an adequate opportunity to sleep, which can lead to daytime consequences, such as fatigue, difficulty concentrating, and irritability.(5),(6) Insomnia is one of the most common sleep-wake disorders. Approximately 30% of adults worldwide have symptoms of insomnia.(7),(8) In particular, older adults tend to have a higher prevalence rate with many experiencing insomnia symptoms for months to years. As a result, insomnia causes various social losses, such as long absences and reduced productivity. It can increase the risk of falling in older adults.(9)
Through the launch of DAYVIGO, Eisai will continue to prioritize the provision of appropriate usage and safety information. By providing DAYVIGO as a new option for the treatment of insomnia, Eisai aims for contribution to the restoration of daytime function and recovery for patients with insomnia by delivering an active daytime life through fast sleep onset and good quality sleep.
For more information visit https://www.eisai.com/news/2020/pdf/enews202036pdf.pdf.
About Eisai
Eisai Co., Ltd. defines our corporate mission as "giving first thought to patients and their families and to increasing the benefits health care provides," which we call our human health care (hhc) philosophy. With approximately 10,000 employees working across our global network of R&D facilities, manufacturing sites and marketing subsidiaries, we strive to realize our hhc philosophy by delivering innovative products to address unmet medical needs, with a particular focus in our strategic areas of Neurology and Oncology. As a global pharmaceutical company, our mission extends to patients around the world through working with key stakeholders to improve access to medicines in developing and emerging countries.
For more information about Eisai Co., Ltd., please visit https://www.eisai.com
Contact:Public Relations Department Eisai Co., Ltd. +81-(0)3-3817-5120
Copyright 2020 JCN Newswire. All rights reserved. www.jcnnewswire.com
Authors: ACN Newswire - Press Releases
Read more //?#